Dr. Hollenbeck and Devireddy Publish in Journal of Neuroscience
07-30-2015
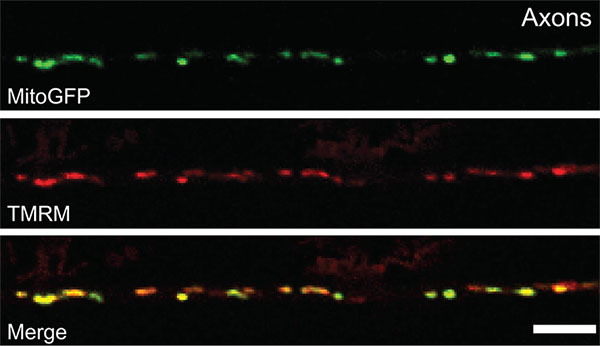
Mitochondria, the organelles within our cells that produce chemical energy from foodstuffs, have their own life cycle: they grow, divide, fuse and when they wear out they are turned over. In neurons – enormous, energy-hungry, compartmentalized cells – it is now clear that mitochondrial turnover is important in neurodegenerative disease. A recent study from the Hollenbeck laboratory used mutations in a Parkinson's disease-related protein, PINK1, to show that current ideas about how mitochondria turn over, and how this process fails in Parkinson's disease, are probably oversimplified and incorrect. Using quantitative in vivo measurements in the intact Drosophila nervous system, Devireddy et al showed that while deleting the PINK1 gene produced the expected, disease-like defects in mitochondrial metabolism, it did not cause the accumulation of senescent, undigested mitochondria in the axons of motor nerves. Instead, PINK1 deletion produced defects in the ability of mitochondria to both enter and exit the axon, along with abnormal mitochondrial size and shape in the motor cell bodies only. These data argue for a restriction of mitochondrial turnover to the cell bodies, and a more subtle and compartmentalized effect of Parkinson's disease pathology on neuronal mitochondria. Swathi Devireddy, a graduate student in the lab, performed this study, with assistance from BIOL undergraduates Alex Liu and Taylor Lampe.
Devireddy, S., Liu, A., Lampe, T. and P. J. Hollenbeck. 2015. The organization of mitochondrial quality control and life cycle in the nervous system in vivo in the absence of PINK1. Journal of Neuroscience 35:9391-9401.