A Fatty Sandwich in the Membrane
07-11-2014
Structures of complex membrane proteins occupy less than 1% of structures deposited in the Protein Data Base. Studies on a multi-component photosynthetic membrane protein complex provide an expanded perspective of the interactions of membrane lipids with integral membrane proteins. Lipids are seen to not only provide the membrane environment for proteins embedded in membranes, but as seen in a high resolution structure of the cytochrome complex that is central to photosynthesis, lipids also function in the internal protein structure. The protein complex contains 23 lipid-binding sites in the monomeric unit of the complex, which mediate crosslinking of the subunits in the complex, stabilize the dimeric structure, and are proposed to mediate formation of "super-complexes." Another consequence of the intra-protein lipid is variation through the protein complex of the properties associated with the storage of electrons ("dielectric heterogeneity"). The lipid-protein studies were carried out by then-graduate student, S. S. Hasan in the lab of W. A. Cramer, and the studies on dielectric constants by Hasan, Research Associate Dr. S. D. Zakharov in a collaboration of the Cramer lab with the lab of S. Savikhin and then-grad student, A. Chauvet in the Physics Dept.
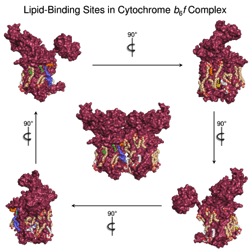
Internal Lipid Architecture of the Hetero-Oligomeric Cytochrome b6f Complex. Hasan, S. S. and W. A. Cramer. 2014. Structure, 22:1008-1015.
A Map of Dielectric Heterogeneity in a Membrane Protein: The Hetero-Oligomeric Cytochrome b6f complex. Hasan SS, Zakharov SD, Chauvet A, Stadnytskyi V, Savikhin S, and Cramer WA. 2014. J Phys Chem B. 118: 6614-25.
Article and photo provided by William Cramer, Henry Koffler Distinguished Professor of Biological Sciences.