Jue Chen
01-07-2010

Associate Professor Biological Sciences. Investigator, Howard Hughes Medical Institute
ATP-binding cassette (ABC) transporters are multi-domain proteins that pump a variety of substrates across biological membranes. More than a dozen human diseases have been traced to ABC proteins including cystic fibrosis, the most prevalent monogenetic disease in Europe and the Americas. Over-expression of some ABC transporters in tumor cells confers drug resistance by pumping the drugs out of the cells, a phenomenon that becomes one of the major barriers to effective treatment of cancers.
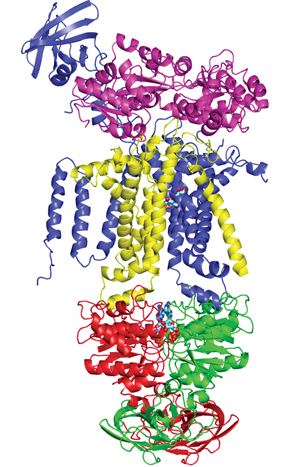
The most remarkable features of ABC transporters include: (1) the substrates transported by different members of the ABC superfamily are largely diverse, arranging from small ions to large polypeptides and polysaccharides. To understand the basis of the selectivity from one ABC transporter to another is a major challenge. (2) the movement of the substrate, powered by ATP binding and hydrolysis, can be outward (export) or inward (import), which means that the substrate can be taken up by the transporter from either side of the membrane. It remains elusive whether exporters and importers share a common mechanism of action. We use X-ray crystallographic and biochemical approaches to address these questions.