Donald Ready

Research Areas
My lab has focused on pattern formation and differentiation in the developing Drosophila compound eye. The ability to manipulate fly eyes using genetic, molecular and cellular tools has made them a valuable model system for the dissection of highly conserved developmental pathways. During pattern formation, an initially unpatterned epithelium is transformed into reiterated clusters of 8 photoreceptors, future unit eyes or ommatidia, surrounded by a precise network of supporting cells. Upon completion of pattern formation, when all retinal cells have been allotted their fate and established their appropriate, specific cell-cell contacts, differentiation begins. Most spectacularly, photoreceptors undergo enormous growth and build a rod-like photosensitive membrane organelle, the rhabdomere. Supporting cells likewise execute specific differentiative programs to build lenses and shape ommatidia to their function. In addition to optically insulating ommatidia, the hexagonal network of inter-ommatidial pigment cells plays a critical role in aligning rhabdomeres to the unit eye’s optical axis and aligning the eye’s photoreceptor array to give the fly its panoramic view, which makes them so hard to catch.
In collaboration with Dr. Henry Chang, Dept. of Biological Sciences, my current focus is how intercellular calcium waves, the actomyosin cytoskeleton, and extracellular matrix cooperate in compound eye morphogenesis.
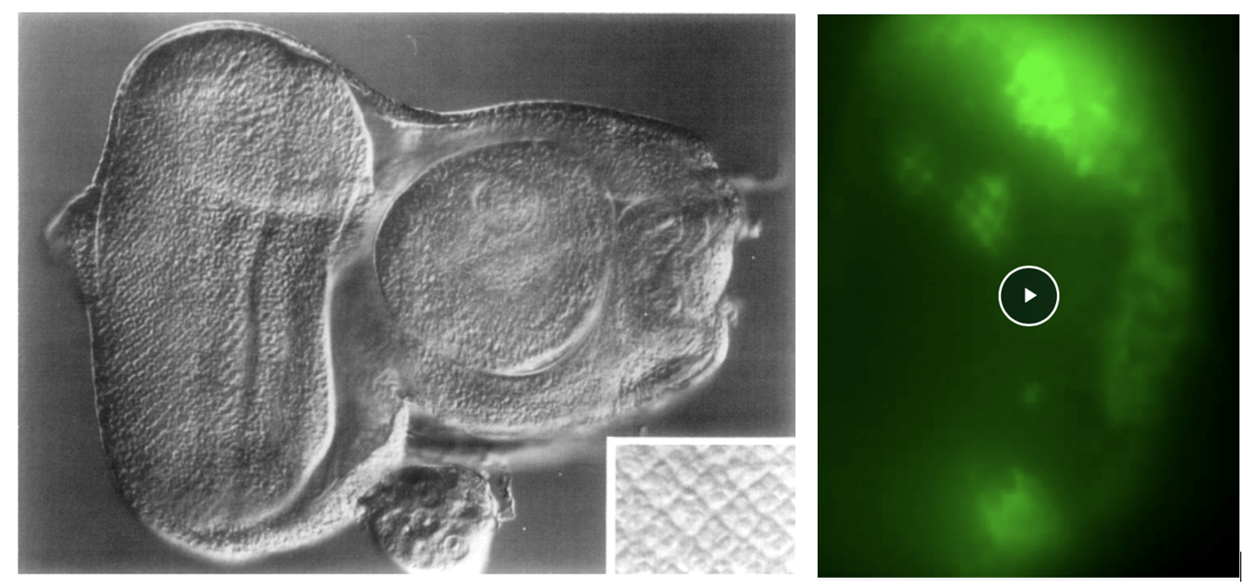
Left : Ready, Hanson and Benzer (1976), Fig. 7, shows an eye-antennal disc. Mature photoreceptor clusters form a square array at the posterior of the eye disc (left and inset). Anterior to the clusters, a vertical dark line, the morphogenetic furrow, marks the onset of pattern formation.
Right : Ready and Chang (2021), Video-5, shows time lapse GCaMP6 imaging of calcium waves in the developing retinal epithelium. To the left, posterior, waves sweep across the grid of supporting cells surrounding developing photoreceptor clusters. Right of center, the bright, vertical band of activity marks the morphogenetic furrow. http://movie.biologists.com/vi...
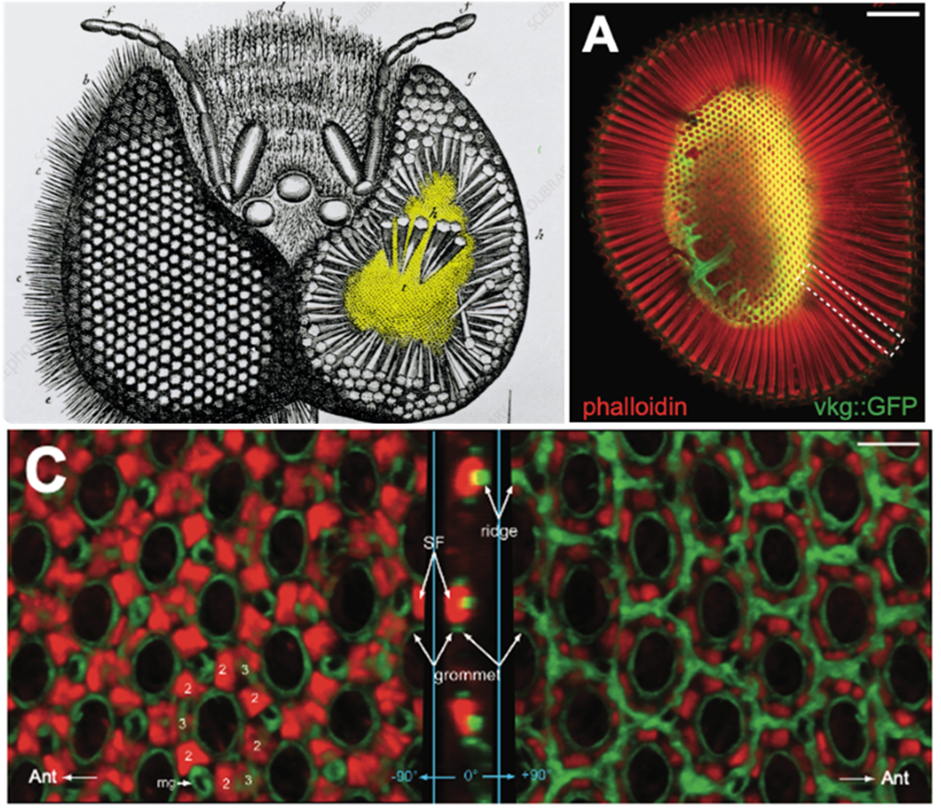
Top left : Jan Swammerdam (1650). Honeybee eye dissection. Ommatidia, tapered hexagonal prisms, stand on a perforated, domed retinal floor (yellow).
Top right and bottom : Ready and Chang (2021), Fig. 1. A. Actin-rich, photosensitive rhabdomeres, boxed, extend from the outer, corneal surface of the eye to the retinal floor (yellow), a cytoskeletal/ECM assembly shaped by Actin (red) and Collagen IV/Viking (green). C. The retinal floor viewed from above (left panel), side (middle panel), and below (right panel). Actin stress fibers (SF) in inter-ommatidial cell endfeet anchor to neighboring grommets, ECM perforations via which ommatidial axons exit the retina, and pull them towards each other, contracting the floor fourfold and imposing a taper upon overlying ommatidia. A hexagonal network of Collagen IV ridges resists compression. A balance of actomyosin contraction and Collagen IV stiffness shapes the curvature of the retinal floor, aligning ommatidia to the eye’s panoramic view. Intercellular calcium waves coordinate stress fiber contraction across the eye.
Education
Ph.D., California Institute of Technology, 1976